Hello so this page should hopefully include some useful information on chemistry, physics and things that might come up more than once, its a bit difficult to summarise and there will definitely be better resources than my ramblings for you to use! but if youre here reading i hope you enjoy :)
# Intermolecular forces
For some context, electrons are often depicted as being localised around one atom evenly in "electron shells" they're often localised to the molecule itself and seemingly not impacted by the the bonded atoms properties. However, even in a net neutral molecule like water the relative properties of atoms in the molecule can impact electron movement, bond forming and interactions.
some atoms have high **electronegativity** which means their propensity to attract electrons is high, thus if you have a molecule of water the oxygen with high electronegativity will attract the electron from the hydrogens.
All of this is to say that, while it may be useful to visualise electrons as being in discrete electron shells, when other atoms are involved their behaviour can vary drastically.
### Hydrogen bonding
this is a classsiccc!!! if you ever wondered why ice floats in water when solids are supposed to be more dense than liquids Hydrogen bonding is why!
It is the strongest of the transient intermolecular forces,
using water as an example as stated above, oxygen has a high electronegativity and attracts electrons from its hydrogens this results in the oxygen in the molecule of water being slightly more negatively charged and the hydrogens being more positively charged.
this then allows for attractive forces between water molecules to occur between the hydrogen of one molecule to the oxygen of another. this interaction is not limited to only hydrogen and oxygen but hydrogen that is covalently bonded to any atom with high electronegativity.
the reason these are so strong is because of the small size of hydrogen
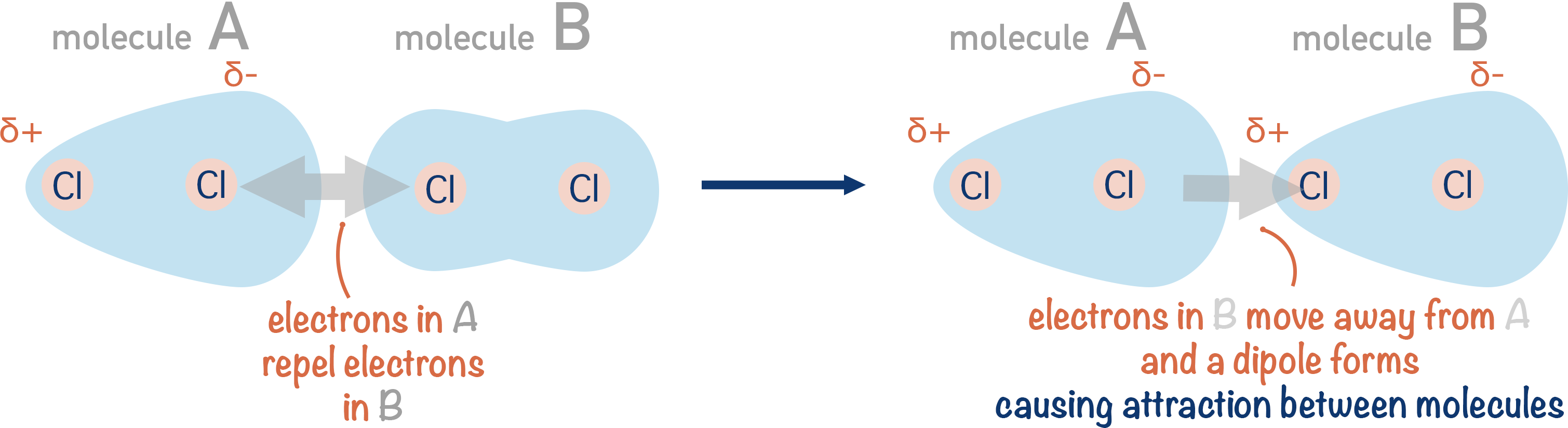
### Dipole-Dipole
much like hydrogen bonds (h-bonds essentially being a dipole-dipole) the atom with high electronegativity that is bonded covalently to another atom pulls away the electrons creating a partial positive charge in one atom and itself become partially negative. in this case the atom bonded to the atom with high electronegativity can be anything (within the constraints of being covalently bonded). these interactions can now occur as attractive forces between partially negative and partially positive pairs.

### Induced dipole dipole
AKA london forces, these occur between all molecules as distribution of electrons constantly changes as they move around. sometimes the movement can cause an "induced dipole moment" in which the electrons become more concentrated at one side of an atom or pair of atoms in a bond temporarily. this then causes a dipole which the opposing transient charges can interact between molecules or atoms.
# Entropy and the second law of thermodynamics.
Okay so I think this will be a bit long but I really want to walk you through this because I actually think a lot of places focus on the core values of entropy and thermodynamics but blindside you with so much related jargon that it becomes a case of having to constantly find out what things theyre talking about mean. I cant guarentee this will be completely entry level however since this is something I too struggle to understand i hope I can try and put it into words that make sense to me, so hopefully they will make some sense to you :) however this doesn't actually come up all that much in the doc so feel free to skip :p
**Entropy** is a measure of the dispersal of energy in a system, or how spread out and inaccessible it is for that energy to do useful work.
In thermodynamics, “useful work” refers to the portion of energy that can be harnessed to do mechanical work. Not all energy transformations are 100% efficient some energy is inevitably lost as heat, which increases the entropy of the system. **Mechanical work** is the transfer of energy that happens when a force is applied to something and it moves in the direction of that force. It’s basically how we convert energy into motion In thermodynamics, mechanical work often involves gases expanding or being compressed in systems like pistons and the gas does work when it expands and pushes the piston out, or work is done **on** the gas when it’s compressed. mechanical work is organised and coordinated which contrasts thermal energy which spreads out more randomly (which is why loss of energy as heat increases entropy of a system; but specifically because that heat becomes dispersed energy, increasing the number of accessible microstates in the system and surroundings.).
I will also just touch on briefly the second law of thermodynamics which **that the total entropy of an isolated system can never decrease over time.**
It can stay the same (in ideal, reversible cases), but in real-world processes, **it always increases.** Thermal energy will disipate and cooler surrounding objects will increase in thermal energy eventually there will be equalibrium in the system (Ie everything having the same amount of thermal energy). However, cooler objects can not, without work put in, transfer their thermal energy to objects already of higher thermal energy.
When calculating chnages in entropy the following is used:
$ΔS=qrev/T$
T is temperature in kelvins
This only accounts for entropy change in an idealised system where we consider the heat added as reversible (qrev). In thermodynamics, **reversible heat** means **heat transfer that happens infinitely slowly**, in such a controlled way that the system is always just barely out of equilibrium with its surroundings and can be reversed. This also means that we calculate with the idea that no energy is lost from the system or is wasted. irregardless of how messy a real world process is.
**Entropy** is more classically used to describe the degree of disorder in a system. Thermodynamic entropy quantifies the number of microscopic arrangements (microstates) that correspond to a given macroscopic state (macrostate) of a system.
A **macrostate** describes the macroscopic properties of a system, such as its temperature, pressure, or volume. A **microstate** refers to a specific configuration of the system at the microscopic level, such as the positions of all its particles.
Because of the Idea of randomness a system is going to want to adopt some microstate that is a substate of the most common configuration the image below will hopefully explain this;
![[Microstates.jpg]]
In this example the most likely microstate is one of the (C) substates, And because there is the most substates in (C) it also means this configuration has the highest **Entropy** this idea is also factored into how we calculate the entropy of a system
$S=klnW$
where K is the Boltzmann constant: 1.380649 x 10⁻²³ J/K
and W is the number of microstates after a process divided by the number of microstates before a process, if the number of microstates increases that confers an increase in entropy.
### Enthalpy
Enthalpy is defined as the sum of a thermodynamic system's internal energy and the product of its pressure and volume.
$ H=U + pV $
where U is the internal energy of a system and pV are pressure and volume respectively.
Its most beneficial use is calculating change in enthalpy whether heat energy is released or taken in during a process (eg protein folding, ligand binding or complex formation) as it is not possible to directly measure absolute enthalpies.
A process that results in more bonds formed/ interacts after it has taken place is enthalpically favourable and ΔH is negative; this may seem counterintuitive since reactions often require energy to be put in. This energy is however in relation to activation energy not actual thermodynamic principles. Systems want to minimize energy . If a new arrangement (like a folded protein) forms stronger interactions, the system will give up energy (as heat) and become enthalpically favorable.
This concept may also go against entropic favourability (prefering to maximise the number of microstates) however in biological environments we also have to consider the impact of water and "hydrophobic effect". Water often orders itself around a molecule in a cage like structure "clathrates" as they can sometimes be reffered to- this results in decreased entropy. Protein folding and hydrophobic molecules clustering together will diminish the amount of ordered water in a system, by literally reducing the surface area for water to order around- resulting in increased entropy for the water. this means that even though a proteins/molecules entropy decreases the entropy of the water somewhat compensates. The entropy of the system can, in some cases increase overall, even if the protein itself becomes more ordered.
### conformational entropy